Dermatology
We support your dermatological drug development program from early stage candidate screening to clinical testing. We focus on IVRT testing and assess intradermal PK and PD profiles ex-vivo and in-vivo using our propritary open flow microperfusion (OFM).
In Vitro Release Testing (IVRT)
IVRT with Franz diffusion cells in combination with GLP compliant bioanalytical analyses.
Ex-vivo PK Testing in Skin
We investigate and compare skin penetration and release rates of topically applied APls.
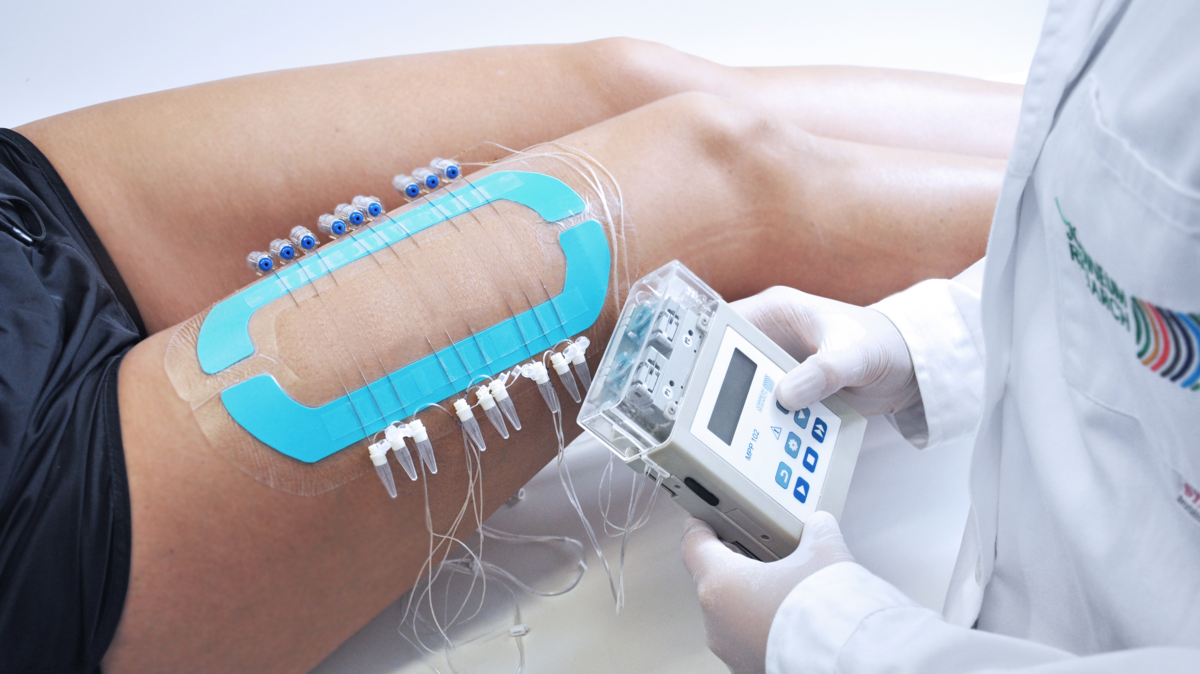
Preclinical Studies in Skin
In-vivo preclinical assessment of intradermal PK/PD profiles in animal disease models.