Our Partnership
Combining the scientific innovations from JOANNEUM RESEARCH and the comprehensive capability of AXIS Clinicals to support clinical development, we believe that this partnership is a crucial milestone to establish dOFM™ as a translational approach that provides unique cutaneous pharmacokinetic data with a proven potential in guiding the development of dermatological drug products toward clinical stage success.
Background
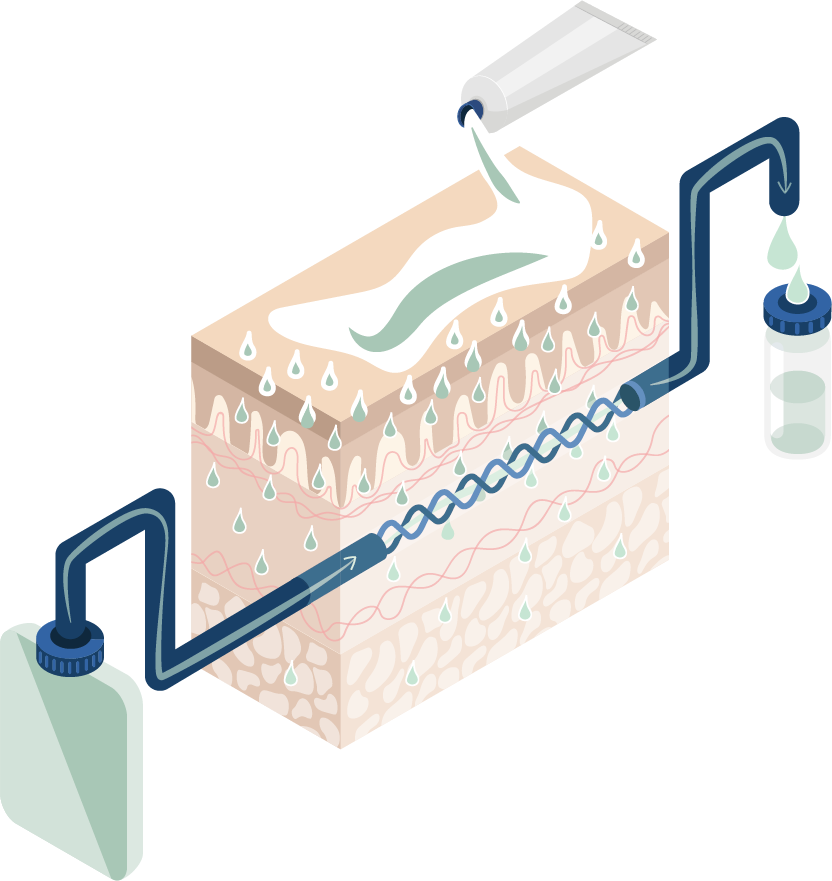
dOFM™ is a proprietary continuous sampling technology in dermal tissue that was developed at HEALTH – Institute for Biomedical Research and Technologies, a research institute at JOANNEUM RESEARCH Forschungsgesellschaft mbH. HEALTH has a demonstrated track record in supporting drug development and adding asset values for pharma companies (more than 19 clinical and 100 preclinical trials conducted to date). Specifically, clinical dOFM™ studies have been conducted to determine PK-PD correlations at or near the site of action to de-risk and endorse later-stage clinical trials and further development decision.
JOANNEUM RESEARCH Forschungsgesellschaft mbH (JOANNEUM RESEARCH) is a successful nationally and internationally active research institution owned by the Austrian federal states of Styria, Carinthia, and Burgenland. It is linked to a worldwide network and has been providing leading research according to the highest international standard since the 1960s. The Institute for Biomedical Research and Technologies (HEALTH) develops comprehensive, interdisciplinary solutions to problems encountered in the fields of medicine, pharmacy, medical technology, and healthcare research. Frank Sinner (VP – JOANNEUM RESEARCH HEALTH) states: “JOANNEUM RESEARCH is looking forward to the close collaboration with AXIS Clinicals US, which will allow expanding the portfolio of unique clinical dOFM services at the highest quality standards. AXIS Clinicals LLC (AXIS) is a privately held contract research organization, established for 20 years, that specializes in topical products’ clinical research. As a global operation with comprehensive clinics and bioanalytical laboratories in United States, India, and Mexico AXIS has been successfully audited by FDA, WHO, and various countries’ regulatory agencies. Dinkar Sindhu (CEO – AXIS Clinicals US) states “AXIS is pleased to be partner with Joanneum Research and include dOFM™ expertise to assist in establishing solid clinical knowledge for all phases of dermal products’ research programs”.

Contact Us
Please reach out and we will answer your questions, react to your ideas and deal with your requirements.